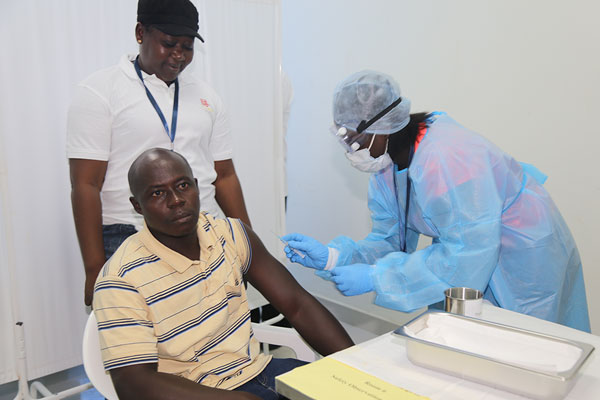
A volunteer in Sierra Leone gets a shot to test the Chinese-developed Ebola vaccine’s safety and effectiveness on Oct 10.[Photo/Xinhua]
China’s first homegrown vaccine against Ebola has entered its second phase in a clinical trial in Sierra Leone in West Africa, marking a breakthrough in overseas testing of Chinese-developed vaccines.
Chen Wei, a researcher at the Institute of Microbiology and Epidemiology of the Academy of Military Medical Sciences, said on Oct 12 that dozens of volunteers recruited from among local people had been given the shots to test the vaccine’s safety and effectiveness.
Her team developed the vaccine against the deadly virus with partner Tianjin CanSino Biotechnology.
The ongoing trial in Africa followed the first phase of testing, which was conducted in China using Chinese and African volunteers, she said.
To bring the trial to Africa, the team went through rigorous reviews of intellectual property rights, technology and ethics, and over the weekend the team was given approval by authorities in Sierra Leone to conduct the trial, Chen said.
“The trial in Africa also helps China gain knowledge and useful experience for future medical cooperation in a global context,” she said.
She didn’t give a timetable for the trial, saying that “depends on the volunteer recruitment as well.”
“The volunteer recruitment is still underway and is running smoothly, since the local people are friendly to Chinese,” she added.
Since March last year, Ebola has infected 28,000 people and killed at least 11,300 in an outbreak mainly in West Africa, including Sierra Leone, according to the World Health Organization. It is now considered to be under control.
The Chinese vaccine candidate is based on the mutant gene type from last year and especially targets the strain that hit West Africa, previous reports have said.
The vaccine is in the form of a freeze-dried powder that will be stable for at least two weeks in temperatures of up to 37 degrees Celsius.
This will make it suitable for the tropical West Africa region and for large-scale production, according to a report based on a review by 17 experts, Xinhua News Agency reported.
China is the third country to put an Ebola vaccine into clinical trials following the United States and Canada.
The Chinese drug has obvious advantages compared with the other vaccines in the clinical phase, the report said.
The other vaccines are based on a gene type from the 1976 outbreak and are in liquid form and must be stored at minus 80 degrees Celsius, it added.
